The post History of #BCSM appeared first on Dr Deanna Attai.
]]>A History of #BCSM and Insights for Patient-Centered Online Interaction has just been published!
The Twitter hashtag #BCSM (breast cancer social media) was first used in July 2011, by patient advocates Alicia Staley and Jody Schoger, when they started a weekly breast cancer-focused chat. The hashtag is currently used not only for the weekly chats, which focus on education and support, but also to tag any information related to breast cancer.
We evaluated the use of the #BCSM hashtag from 2011 – 2019, and the findings included:
- 7500 unique users tweeted using the #BCSM hashtag 830,000 times
- 440,000 tweets were unique (not retweets or quoted tweets)
- There were 4.2 million impressions, an indicator of potential reach or views
Looking at the annual statistics:
- There was an increase in unique users from 602 in 2011 to 19,800 in 2019
- Patient advocate accounts increased from 163 in 2011 to 1018 in 2016 (peak) to 794 in 2019
- Doctor / Healthcare provider accounts increased from 96 in 2011 to 3016 in 2019
Additional statistics are provided in the (open access) manuscript. In the paper, we also discuss some of the challenges to community sustainability, including patients desiring a more focused support community (such as exclusively for Stage 4 or lobular breast cancer, or groups focusing on a specific racial / ethnic group), increasing “noise” due to increased numbers of participants, and moderator burnout. These will be important to address going forward not only for #BCSM, but for other online patient support communities.
#BCSM was the first cancer-specific chat, and has inspired the formation of other communities including for lung cancer, brain tumors, and gynecologic cancers. The hashtag and the community that has developed around it serve as a way to connect patients with each other and with physicians and researchers. Despite the challenges going forward, we have demonstrated that social media can be used as an important source of education and support for anyone impacted by breast cancer.
The post History of #BCSM appeared first on Dr Deanna Attai.
]]>The post Endocrine Therapy Survey Results appeared first on Dr Deanna Attai.
]]>Last year, we asked the online breast cancer community to participate in a survey to assess experiences with endocrine therapy (ET). We are proud to announce that the study has now been published, in the Journal of Cancer Survivorship*.
First of all, I would like to thank all of the participants – we surpassed our accrual goals and this is the largest survey of ET use by patients who participate in online breast cancer communities!
About the respondents:
- 111 respondents did not start the recommended ET, and concern about side effects was the primary reason
- Of those who took ET (2407), 2353 were women and 54 were men
- Most of the women (74%) were post-menopausal
- Mean age at diagnosis was 50 for women (range: 23-82) and 54 for men (range: 24-73)
- Most (87%) were diagnosed at Stage 1-3
- 100 (4.2%) were diagnosed with de novo Stage 4 / metastatic breast cancer
- 12% of those diagnosed at an early stage eventually developed Stage 4 / metastatic breast cancer
Treatment:
- Aromatase inhibitors (AIs) were the most commonly used medication
- 91% of respondents reported at least one class of side effect that they felt was related to treatment (92% of women and 74% of men)
- Musculoskeletal and general physical changes (such as weight gain and unhappiness with body image) were the side effects most commonly reported by women
- Men most commonly reported sexual and cognitive / mood side effects
- 33% (33% of women and 50% of men) discontinued therapy early
- 9% reported that they took treatment breaks or discontinued therapy early either without informing their medical team or against their medical team’s advice
Side effect management:
- 3 classes of side effect management strategies were felt to be most helpful:
- Healthy diet, exercise, physical therapy
- Complementary therapy such as yoga, acupuncture and meditation
- Vitamins, supplements and herbs including medical marijuana
- Only 41% of respondents noted any relief from side effect management strategies
Medical team communication: (multiple responses permitted so this category did not add up to 100%)
- 70% felt supported by their medical team in attempting to discuss side effects
- 32% were made to feel that they should be better able to handle side effects or that the side effects were not related to treatment
- 7% did not discuss side effects with their treatment team, feeling that there were more important issues to discuss, that there was not enough time, or they did not feel comfortable
Some other findings:
- Respondents with early-stage and metastatic breast cancer reported similar side effects and management experiences, even though these two groups of patients have very different supportive needs
- Men who responded to our survey were less likely to report side effects but more likely to discontinue therapy early compared to women – more information is needed about the experience of men with breast cancer and those taking endocrine therapy
Clearly, there is room for improvement in terms of medical team support and understanding. In addition, as only 41% of respondents noted any relief from side effect management strategies, we need more effective treatments for ET-related side effects. Thank you to all who participated in this survey! We are hopeful that your responses and comments will inspire researchers devote more time to addressing these important issues.
*If you are not able to access the full study and would like a copy, please email me: contact at drattai dot com
The post Endocrine Therapy Survey Results appeared first on Dr Deanna Attai.
]]>The post Discordance Between Primary and Metastatic Breast Cancer appeared first on Dr Deanna Attai.
]]>As part of the pathologist’s evaluation of a breast cancer, the presence or absence of the estrogen and progesterone receptor (ER/PR) and level of Her2/neu gene expression is determined. These are important factors used to help guide treatment recommendations for endocrine therapy, chemotherapy, and Her2-targeted therapy.
If metastatic breast cancer develops (when the primary tumor spreads to other areas of the body, such as the lungs, liver, bone, or brain), it is recommended that ER/PR and Her2/neu testing be performed on the metastatic cancer. It is known that cancers can sometimes mutate and the metastatic lesion may have different characteristics compared with the primary lesion. While current international guidelines do recommend biopsy of the metastatic tumor, this is not always performed.
A study recently published in Breast Cancer Research and Treatment reported on discordance between the primary and metastatic breast cancer. The study was performed in Germany. Among patients treated for breast cancer between 1982 – 2018, 541 had receptor status from both the primary and the metastatic breast cancer documented in the medical record.
The researchers found that there was a 14% discordant rate for ER, a 32% discordant rate for PR, and a 15% discordant rate for Her2/neu. All of these were felt to be clinically meaningful, in that a change in ER/PR or Her2/neu status would result in a change in treatment recommendations. If a tumor loses the ER/PR receptor, endocrine therapy (such as tamoxifen or aromatase inhibitors) would be ineffective. If a tumor gains the Her2 protein, Her2-targeted therapy should be instituted.
They noted several reasons for discordance including:
- Variability in the testing process, especially as testing procedures have changed over time
- Tumor heterogeneity – cancers may not be made up of a single cell “clone”
- Mutation of the tumor over time
Median follow up was 58 months. They noted that loss of ER/PR positivity was associated with worse overall survival, and gain in Her2 positivity was associated with improved overall survival. However, one limitation of the study is that they could not determine if the survival differences were actually due to the changes in the tumor and subsequent treatment. They noted other limitations of the study including that the dataset included patients with Her2 positive disease who were treated prior to the approval of trastuzumab (Herceptin), there were changes in cut-off values for what was considered ER/PR “positive” during the study period, and receptors were not re-checked in a centralized lab.
However, despite the limitations, it is important to note that treatment recommendations could be altered as a result of testing the metastatic cancer. The researchers concluded that “Where feasible, metastatic lesions should be biopsied in accordance with current guidelines.” If you have been diagnosed with metastatic breast cancer, it is important to discuss biopsy of the metastatic tumor with your oncologist, and not assume that it is the same cell type as the primary lesion. This study found that in at least 14% of cases, knowledge of the metastatic tumor biology could alter treatment recommendations.
The post Discordance Between Primary and Metastatic Breast Cancer appeared first on Dr Deanna Attai.
]]>The post Cancer Survivorship Care appeared first on Dr Deanna Attai.
]]>The American Cancer Society estimates that as of January 2019 there were 16.9 million individuals in the United States living with a history of cancer, and 1.76 million newly diagnosed with cancer. Traditionally, patients return to their oncology team, primarily their medical oncologist, for follow up care after they complete active treatment. However, there are not enough medical oncologists to care for the anticipated increase in newly diagnosed patients as well as cancer survivors, and the American Society of Clinical Oncology and other organizations recommend shifting more of survivorship care to primary care physicians.
The aims of our study are to determine:
- Which member of the medical team is the primary point of contact for cancer survivorship care
- Which member of the medical team does the patient prefer to be the primary point of contact for cancer survivorship care
- Do patients perceive that their primary care providers are comfortable providing cancer survivorship care
- Would patients be accepting of cancer survivorship care solely from their primary care provider
- Are patient preferences influenced by age, age at diagnosis, type of cancer, treatments received, or severity (stage) of cancer
This survey is being conducted for research purposes. It is a UCLA research study, IRB# 20-001103.
Individuals who have been diagnosed with one or more cancers are eligible to participate.
This research survey should take approximately 20-30 minutes to complete and all responses are anonymous. The anonymous data will be securely stored by the principal investigator and may be used for future research studies. There is no industry funding or sponsor for this study. Results will be shared once data analysis is complete. We appreciate your time and thank you in advance for your participation.
- Deanna J. Attai, MD
- Larissa Nekhlyudov, MD
- Matthew S. Katz, MD
- Beverly A Zavaleta, MD
Survey Link: https://uclahs.az1.qualtrics.com/jfe/form/SV_aa46PeMQzIfJgAB
For questions regarding this study, you may contact the principal investigator Dr. Deanna Attai
- By phone: (818) 333-2555
- By email [email protected]
- Or by mail: 191 S. Buena Vista #415, Burbank, CA 91505
UCLA Office of the Human Research Protection Program (OHRPP):
If you have questions about your rights as a research subject, or if you have concerns or suggestions and you want to talk to someone other than the researchers, you may contact the UCLA OHRPP
- By phone: (310) 206-2040
- By email: [email protected]
- Or by mail: Box 951406, Los Angeles, CA 90095-1406
The post Cancer Survivorship Care appeared first on Dr Deanna Attai.
]]>The post Surgery and Metastatic Breast Cancer appeared first on Dr Deanna Attai.
]]>An abstract presented at last week’s virtual meeting of the American Society of Clinical Oncology focused on whether or not there is a benefit to surgery for the primary breast tumor in patients with metastatic, or Stage IV, breast cancer.
Metastatic breast cancer (Met-BC) is when the breast cancer spreads beyond the breast and underarm lymph nodes. Common sites of metastases include the lungs, liver, bones, and brain. Anyone diagnosed with breast cancer has the potential to develop metastatic disease, but approximately 5-10% of patients are found to have Met-BC at the time of initial diagnosis, known as de-novo Stage IV. For these patients, it has been controversial whether or not to recommend some form of breast surgery (lumpectomy or mastectomy) to remove the primary tumor. Some studies have shown a survival benefit when the primary tumor is removed, but widespread adoption of the results of these studies has been limited because in many of these studies, patients who underwent surgery were younger, healthier, and had a lower volume of metastatic disease compared with the general population of patients with Met-BC.
Dr. Seema Khan presented the results of the ECOG-ACRIN Research Group E2108 study. In this study, patients who had stable Met-BC (after 4-8 months of treatment) were randomized to either surgery or no surgery for the primary tumor. The two study groups were well-matched for age, race, and breast cancer subtype. With a median follow up of 59 months, there was no improvement in overall survival or in progression-free survival. Local progression (cancer continuing to grow in the breast) was more common in patients who did not undergo surgery (25.6 versus 10.2% at 3 years). At 18-month assessment, health-related quality of life was significantly worse in those who underwent surgery, but no differences were observed at the 6-month or 30-month assessment intervals.
During her presentation, Dr. Khan acknowledged that there may be situations where surgery for the primary tumor is a reasonable option in the setting of Met-BC. She noted that there is room for individualization, but stressed that as there is no survival benefit and no improvement in quality of life, surgery for the primary tumor in the setting of Met-BC should not routinely be performed.
It is important to note that any medical meeting abstract, whether an oral presentation or poster, has been evaluated by the meeting program committee but has not been subject to rigorous peer review as would occur with a formal manuscript submission. Abstracts often do not include the full set of results, and additional findings may be included in the eventual publication, some of which may be different than those presented in the abstract. We look forward to the peer-reviewed publication when it is available.
The post Surgery and Metastatic Breast Cancer appeared first on Dr Deanna Attai.
]]>The post Going Flat After Mastectomy appeared first on Dr Deanna Attai.
]]>Going Flat after mastectomy refers to not undergoing breast mound reconstruction after breast removal. Some patients are not candidates for reconstruction or it is not recommended, however many women may choose to “go flat” even when breast mound reconstruction is an option. This past October, we posted a research survey aimed at better understanding motivations to forgo reconstruction and to identify factors associated with postoperative satisfaction among patients who participate in online breast cancer communities.
The abstract and poster are now posted on the virtual meeting platform of the American Society of Breast Surgeons. Our findings include:
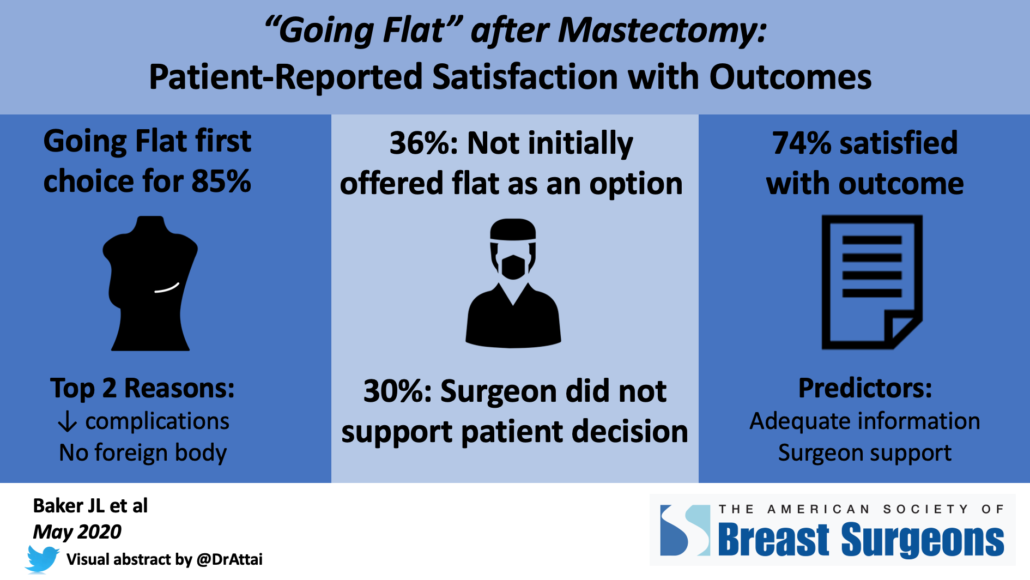
Demographics and timing of going flat:
- 940 women completed the survey, mean age was 53 (range 25 – 84)
- Most patients were white (94%), had private insurance (70%), and were from the US (74%) although 22 countries were represented
- 85% (801/940) did not undergo breast mound reconstruction at the time of mastectomy
- 15% (139/940) initially had reconstruction that was subsequently removed
Top 2 reasons for going flat:
- 72% desire to avoid foreign body such as an implant
- 71% perceived lower complication rate from the surgery
Communication:
- Only 64% were initially offered going flat as an option
- 30% felt that their surgeon did not support their decision to go flat
Satisfaction with outcome:
- 74% agreed or strongly agreed that they were satisfied with their surgical outcome
- Age, race, bra cup size, and history of prior breast mound reconstruction were not associated with postoperative satisfaction
- Strongest predictors of satisfaction were having adequate information about surgical options and surgeon support of the patient’s decision to go flat
Our findings reveal a need for additional research into factors that impact patient satisfaction as well as for surgeon education on how to optimally support women who are not interested in breast mound reconstruction.
It is important to note that any medical meeting abstract, whether an oral presentation or poster, has been evaluated by the meeting program committee but has not been subject to rigorous peer review as would occur with a formal manuscript submission. Abstracts (including ours) do not include the full set of results. It is not unusual for additional findings to be included in the eventual publication, some of which may be different than those presented in the abstract. However, we feel that these results are an important starting point for better understanding of patient motivations for going flat, and also point to a need for improved communication on the part of surgeons. We are in the process of completing a full analysis of the data and look forward to sharing the peer-reviewed publication when available.
On behalf of my co-authors, we would like to thank all who shared and participated in the survey!
The post Going Flat After Mastectomy appeared first on Dr Deanna Attai.
]]>The post When to Stop Screening Mammograms? appeared first on Dr Deanna Attai.
]]>A study published in the Annals of Internal Medicine addresses the use of screening mammography in older women*. The goal of screening mammography (performed on a woman with no suspicious abnormality) is to reduce the likelihood of death from breast cancer. However, there are no randomized studies demonstrating this benefit in older women – they simply were not included in the early studies of screening mammography. Some national guidelines, including from the US Preventative Services Task Force, do not recommend screening mammography past age 75, noting that “the current evidence is insufficient to assess the balance of benefits and harms of screening mammography in women aged 75 years or older.” Some organizations, such as the American Society of Breast Surgeons and the American Cancer Society, recommend that screening mammograms continue as long as life expectancy is at least 10 years. In their Choosing Wisely statement, the American Geriatrics Society recommends that screening tests be considered in the context of life expectancy and take into account the risks of testing, overdiagnosis and overtreatment.
This study used data from Medicare to compare death rates in women age 70 – 84 who underwent screening mammography versus those who stopped screening. Patients had no personal history of breast cancer and had a life expectancy of at least 10 years.
There were slightly more breast cancers detected in the screening group, as expected: 5.5% in the women who underwent screening mammograms versus 3.9% in the women who did not. However, the researchers found that screening did not improve the likelihood of death from breast cancer. Among women age 70-74 there was 1 fewer breast cancer related death per 1000 women, over a period of 8 years, and this was not statistically significant. Among women age 75-84, there was no difference in breast cancer related deaths.
The authors concluded that screening mammography in older women does not significantly impact the likelihood of death from breast cancer. Limitations of this study include that it was an observational study, using information already in a database. The researchers could not adjust for certain factors including use of hormone replacement therapy, family history of breast cancer, and age at first pregnancy, which may impact breast cancer risk.
A separate study* looked at the use of a decision aid in patients age 75 and older. The decision aid provided information about the risks and benefits of screening mammography. In this study of 546 patients, it was shown that receipt of the decision aid prior to an appointment with their physician resulted in women being more knowledgeable about screening mammography and having more discussions with their doctor about screening. In addition, 9% fewer women opted to undergo a screening mammogram. The authors concluded that the use of a decision aid may help reduce over screening in older women.
In the accompanying invited commentary, Drs. Elmore and Ngo-Metzger noted that in women age 74 with average health and life expectancy, screening mammograms prevent approximately 1 death per 1000 exams. However, 1000 women being screened can result in 79 – 96 of them experiencing a false positive (“false alarm”). They pointed out the challenges of screening mammography in older women in that the reduction in death from breast cancer can take many years, (during which the woman may die from other causes) but the potential harms are more immediate.
These two studies and the editorial raise important questions regarding the routine recommendation for screening mammography in older women. The decision whether or not to continue screening should be based on breast cancer risk as well as general health and risk of severe illness or death from other causes. When to stop screening mammography is a challenging, but important conversation to have with our older patients.
- Additional Information:
- Annals of Internal Medicine article summary for patients
- New York Times Aging out of the Mammogram
- E-Prognosis mammogram decision aid, women age 75 – 84
- E-Prognosis mammogram decision aid, women age 85 and older
*If you are not able to access the full study and would like a copy, please email me: contact at drattai dot com
The post When to Stop Screening Mammograms? appeared first on Dr Deanna Attai.
]]>The post Cancer Treatment During COVID-19 appeared first on Dr Deanna Attai.
]]>I was asked to help the UCLA Jonsson Comprehensive Cancer Center team develop some infographics to help answer some common questions that patients with cancer may have regarding their treatment, office visits, and risk of infection. As always, good communication with your medical team is important – so reach out and ask if you have questions or concerns.
The post Cancer Treatment During COVID-19 appeared first on Dr Deanna Attai.
]]>The post Trodelvy for Metastatic TNBC appeared first on Dr Deanna Attai.
]]>A new drug was recently approved by the US Food and Drug Administration (FDA) for the treatment of metastatic breast cancer – breast cancer that has spread to other areas of the body, such as the bones, liver, lungs or brain. Sacituzumab govitecan (Trodelvy), which is given by intravenous infusion, was granted accelerated approval to treat metastatic triple negative breast cancer in patients who have already been on 2 prior treatments for their disease.
Triple negative breast cancer accounts for approximately 15% of breast cancers. It is more common in younger patients, African Americans, and those with a BRCA1 gene mutation. It can be very challenging to treat since there is no specific cellular target, such as the estrogen receptor or Her2/neu protein. Patients with triple-negative breast cancer very commonly receive chemotherapy but the disease may come back or progress despite aggressive treatment.
Sacituzumab govitecan is an antibody-drug conjugate, which is an antibody that is combined with a chemotherapy drug. The antibody targets a protein (trophoblast cell surface antigen-2, or trop-2) on the surface of the cancer cell, and the chemotherapy drug causes breaks in the tumor cell DNA, which leads to cancer cell death. The FDA approval was based on a non-randomized (meaning all patients received the study drug) study of 108 patients who were experiencing disease progression despite receiving between 2 – 10 prior treatment regimens. Among the study participants, approximately 33% experienced a partial or complete tumor response to the medication. Median duration of treatment response was nearly 8 months. Severe adverse reactions included diarrhea, neutropenia (low white blood cell counts), anemia (low red blood cell counts), fatigue, nausea and vomiting. 2 patients stopped treatment due to side effects.
Any advance in the treatment of metastatic breast cancer is welcome news. It is hoped that as new drugs are developed, a longer-lasting response to therapy will be achieved.
Additional Information:
The post Trodelvy for Metastatic TNBC appeared first on Dr Deanna Attai.
]]>The post Tucatinib Approved for Metastatic Breast Cancer appeared first on Dr Deanna Attai.
]]>The US Food and Drug Administration (FDA) has recently approved a new treatment for patients with metastatic breast cancer. Tucatinib (Tukysa) was approved for use in combination with 2 other agents, trastuzumab (Herceptin) and capecitabine (Xeloda) in patients with unresectable (too advanced to remove with surgery) or metastatic (spread to other areas of the body, such as bone, liver, lungs or brain) Her2/neu over-expressed breast cancer (Her2+). Tucatinib is approved for patients who have already been treated with one or more anti-Her2/neu treatments.
Tucatinib is a an oral (pill) medication known as a tyrosine kinase inhibitor. Results of the Her2CLIMB trial were presented in December at the San Antonio Breast Cancer Symposium. The study enrolled 612 patients with metastatic or unresectable Her2+ breast cancer who had received at least one other Her2+ targeted agent but were experiencing progression of disease. Unique to this study was that almost half of the participants had metastases to the brain – often these patients are excluded from clinical trials.
Some of the key findings were that in patients who received tucatinib with trastuzuab and capecitabine versus those who received trastuzumab and capecitabine alone:
- Median progression free survival was 7.8 months versus 5.6 moths
- Median progression free survival for patients with baseline brain metastases was 7.6 versus 5.4 months
- Median overall survival was 21.9 versus 17.4 months
- Confirmed objective response rate was 40.6% versus 22.8%
Serious adverse reactions occurred in 26% of patients and 6% of patients had to stop treatment due to adverse reactions. The most common side effects were diarrhea, hand-foot syndrome, nausea, fatigue, liver toxicity, mouth sores, decreased appetite, abdominal pain, headache, anemia and rash.
The approval of tucatinib provides another option for patients with aggressive breast cancer, including those who have brain metastases. However, as previously discussed, this study is a reminder of how far we have to go to achieve a reliable and long-lasting treatment response in patients with metastatic breast cancer.
Additional Information:
The post Tucatinib Approved for Metastatic Breast Cancer appeared first on Dr Deanna Attai.
]]>