26 February 2019
The US Food and Drug Administration (FDA) has issued a safety communication regarding the use of breast thermography as a sole means of breast cancer screening or diagnosis.
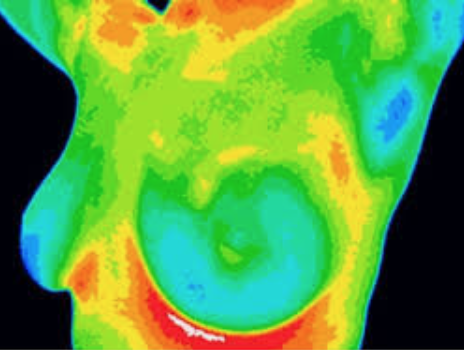
Thermography has been around for many years, and as seen by the FDA communication, has been the subject of multiple warning letters. Thermography works by detecting patters of heat flow on or just below the body surface. We know that breast (and other cancers) tend to have a more robust blood supply compared to normal tissue – and proponents of thermography feel that this reliably translates into detectable changes in heat patterns.
Thermography does not utilize radiation and there is no compression of the breast, making it appear to be an ideal alternative to mammography. Unfortunately, it has never been shown to reliably detect breast cancer, even in patients with suspicious mammography findings (see Collette, AE et al. paper referenced below).
The FDA alert notes that thermography was never approved as a stand alone test to screen for breast cancer, even though many facilities advertise it as an alternative to mammography and other imaging. Thermography practitioners also claim that precancerous changes can be detected many years prior to the development of cancer. I’ve seen many patients over the years who have had abnormal thermograms – and they leave the facility with recommendations including hormone creams (to apply to the breast), lymphatic massage, and repeat thermography in 3 months. Some of these patients in fact had large, advanced cancers. Thermography is not covered by insurance, and costs can range anywhere from $150 – 500 / session. While every thermogram report I’ve seen includes a recommendation for additional imaging with mammogram, ultrasound or MRI (usually in the fine print), this is never really discussed with the patient.
Mammograms have their flaws and early detection does not necessarily result in cure. However, thermography is not the answer.