9 June 2015
Surgery doesn’t help women with early-stage breast cancer – that’s certainly a headline that will get attention. The recent NPR article referred to a study published in JAMA Surgery: Survival Benefit of Breast Surgery for Low Risk Ductal Carcinoma In Situ – A Population-Based Cohort Study(1). The study raises some very interesting points, but the NPR headline is misleading. Early stage breast cancer can refer to Stages 0, I, and 2, and the study cited only refers to low grade ductal carcinoma in situ.
In this study, researchers used the SEER database to identify fifty seven thousand cases of DCIS treated in the United States from 1988-2011. 2% of that group did not undergo surgery. The researchers evaluated breast cancer specific survival in the patients treated with and without surgery in relation to tumor grade. They concluded that there was no survival advantage to undergoing surgery in cases of low grade DCIS. For patients with intermediate grade DCIS, 10 year breast cancer specific survival rates were 98.6% in the group who underwent surgery vs 94.6% in the non-surgical group. For patients with high-grade DCIS, 10 year breast cancer specific survival was 98.4% in the surgical patients vs. 90.5% in the non-surgical group.
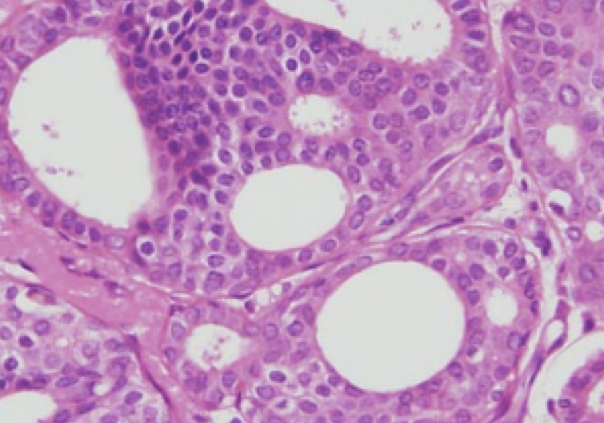
Ductal carcinoma in-situ is also referred to as noninvasive, or Stage 0 breast cancer. It is primarily diagnosed by screening mammogram, as it often does not form a palpable lump. DCIS accounts for approximately 20% of all breast cancers detected by mammography. As screening mammography has become more prevalent, the rate of DCIS detection has increased. Since DCIS does not always progress to invasive cancer, it is a very reasonable for a newly diagnosed woman to ask “Do I need surgery?”.
A hallmark of cancer is the ability to invade surrounding organs and metastasize, and whether or not DCIS should even be considered “cancer” has been the subject of much debate. Dr. Laura Esserman and others have suggested that DCIS be re-classified as an Indolent Lesion of Epithelial Origin(2). The traditional therapy for DCIS is surgical excision (lumpectomy or mastectomy depending on the extent of disease), radiation therapy, and hormonal therapy such as tamoxifen if the DCIS is estrogen receptor positive. The concern of Dr. Esserman and many others is that we are overdiagnosing and overtreating many women. It is estimated that approximately 25-50% of cases of DCIS will likely progress to invasive disease – 60% over 10 years for high grade vs 16% for low grade (1). Preventing invasive disease, which carries a possibility for metastasis, is the primary goal when treating DCIS.
Unfortunately, we are not yet in a position to accurately predict which cases of DCIS will progress and which will not. The study by Sagara et al categorized the DCIS by tumor grade, and this is an important factor in predicting biologic behavior. However, as was pointed out by Margenthaler and Vaughan in their commentary No Surgery for Low Grade Ductal Carcinoma In Situ? (3), a detailed tumor genomic analysis such as the 12-gene assay provides more comprehensive information about tumor behavior and prognosis. Currently this assay is being used in selected cases to classify DCIS as low, intermediate and high risk and to guide treatment. Another limitation of the Sagara study is the retrospective nature, so that information regarding surgical margins and other factors known to be important in recurrence rates is not known. In addition, only 2% of the patients with DCIS underwent non-operative therapy, so the sample size is very small. It is also not known why some women did not undergo surgery.
An additional concern regarding nonoperative therapy is that if surgery is not performed, the diagnosis depends on the accuracy of the core biopsy. In approximately 15-20% of cases when DCIS is found on core biopsy, the surgical pathology actually demonstrates invasive cancer (4). As the entire lesion cannot be sampled with needle biopsy, we don’t know if we are actually observing an invasive cancer.
So can surgery be avoided in women with early stage breast cancer? My answer is in selected cases possibly, but more information is needed. Several ongoing trials will hopefully provide some answers. In the United States, the ALLIANCE trial involves treating patients with letrozole for 3-6 months prior to surgery with tumor assessment by biopsy and MRI. A similar study is being performed at the University of California San Francisco using either tamoxifen or letrozole prior to surgery. Both studies are evaluating tumor biomarkers to help determine if response can be predicted based on specific tumor factors.
In Europe, 2 non-operative trials are opening – LORD and LORIS. Both will include patients with low-grade DCIS and randomize them to either active surveillance or treatment.
We are anxiously awaiting the results of these studies. Identifying women who do not benefit from treatment is an important question that needs to be answered. However at this time, we do not have enough information to make the general recommendation of active surveillance for all women with low grade DCIS.
References:
1. Sagara Y, et al. Survival Benefit of Breast Surgery for Low Grade Ductal Carcinoma In Situ: A Population-Based Cohort Study. JAMA Surg Published online June 03, 2015.;():. doi:10.1001/jamasurg.2015.0895.
2. Esserman LJ, et al. Overdiagnosis and Overtreatment in Cancer: An Opportunity for Improvement. JAMA 2013:310(8)797-798
3. Margenthaler JA, Vaughan A. No Surgery For Low-Grade Ductal Carcinoma In Situ? JAMA Surg Published online June 03, 2015. doi:10.1001/jamasurg.2015.0895
4. Kumiawan ED et al. Risk Factors for Invasive Breast Cancer when Core Needle Biopsy Shows Ductal Carcinoma In Situ.
Arch Surg 2010;145(11)1098-1104







Trackbacks & Pingbacks
[…] help interpreting this confusing finding, especially since Dr. Attai has focused on the issue of surgery and DCIS on her own blog. Dr. Attai emphasized that women don’t die from cancer in the breast, even if […]
[…] help interpreting this confusing finding, especially since Dr. Attai has focused on the issue of surgery and DCIS on her own blog. Dr. Attai emphasized that women don’t die from cancer in the breast, even if […]
[…] help interpreting this confusing finding, especially since Dr. Attai has focused on the issue of surgery and DCIS on her own blog. Dr. Attai emphasized that women don’t die from cancer in the breast, even if […]
[…] help interpreting this confusing finding, especially since Dr. Attai has focused on the issue of surgery and DCIS on her own blog. Dr. Attai emphasized that women don’t die from cancer in the breast, even if […]
[…] help interpreting this confusing finding, especially since Dr. Attai has focused on the issue of surgery and DCIS on her own blog. Dr. Attai emphasized that women don’t die from cancer in the breast, even if […]
[…] help interpreting this confusing finding, especially since Dr. Attai has focused on the issue of surgery and DCIS on her own blog. Dr. Attai emphasized that women don’t die from cancer in the breast, even if […]
[…] help interpreting this confusing finding, especially since Dr. Attai has focused on the issue of surgery and DCIS on her own blog. Dr. Attai emphasized that women don’t die from cancer in the breast, even if […]
[…] more questions regarding appropriate treatments for ductal carcinoma in-situ (DCIS). I wrote about surgery for DCIS about a month ago. The controversy […]
Comments are closed.