20 March 2017
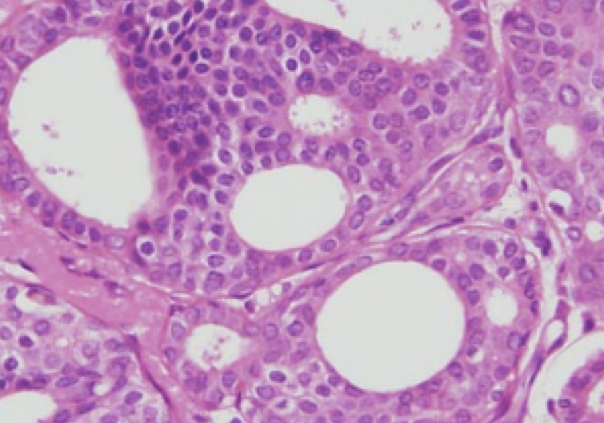
Over 50,000 women in the US are diagnosed every year with ductal carcinoma in-situ (DCIS), also known as Stage 0 breast cancer. DCIS is most often diagnosed on screening mammography and usually presents as a cluster of calcium deposits rather than a lump. In cases of DCIS, malignant appearing cells grow within the milk duct, but do not invade through the wall of the milk duct. DCIS is considered to be a “non-obligate precursor” to invasive breast cancer – it has the potential to develop into invasive disease, but this does not happen in all cases. However, we traditionally have treated DCIS and invasive cancer in a similar fashion – with surgery, radiation therapy and endocrine therapy. It has become clear over the past several years that low grade DCIS is likely a different disease compared to high grade DCIS. Aggressive treatment of low grade DCIS may not improve outcomes but has the potential to cause significant harms.
A new clinical trial has opened for patients with low risk DCIS. The COMET Trial (Comparison of Operative to Monitoring and Endocrine Therapy) is currently enrolling patients under the direction of Dr. Shelley Hwang, a breast surgical oncologist at Duke University. Eligible patients will be randomized to guideline concordant care (standard therapy) versus active surveillance. The primary objective is to evaluate the rate of invasive breast cancer development in the active surveillance group. Secondary objectives include assessments of quality of life and anxiety. In addition, clinical outcomes including mastectomy and breast conservation rates, overall survival and breast cancer specific survival, and ipsilateral (same side) invasive breast cancer rates will be assessed.
The COMET study is not the first to evaluate non-operative therapy for DCIS. The LORIS and LORD trials are already enrolling patients in Europe. A UK-funded initiative known as PRECISION (Prevent Ductal Carcinoma In Situ Invasive Overtreatment Now), headed by the Netherlands Cancer Institute, will collaborate with all three trials.
It is always challenging to go against standard treatment, especially when that means less treatment. It took almost 50 years after the death of Sir William Halsted (“Halsted Radical” mastectomy) for surgeons to accept less invasive surgical procedures. Many patients (and physicians) may feel uncomfortable not removing cells that have been labeled “cancer”. It is important to recognize that lack of surgery does not mean no care – “active surveillance” is now an accepted management strategy for some cases of low grade prostate cancer. Our treatments come with real long term side effects and toxicity. The COMET study is a step in the right direction to help determine which patients may safely avoid aggressive treatment.
Additional Information:
DCISOptions.org
DCIS, Continued…
Slideshare – Are We Overtreating DCIS?
CBS News: Dr. Laura Esserman – When is it OK Not to Treat Cancer?
HealthNews Review Podcast: Dr. Laura Esserman – The DCIS Dilemma
HealthNews Review Podcast: Active Surveillance for Prostate Cancer




Trackbacks & Pingbacks
[…] The next session was Emerging Strategies in Breast Cancer Care, which focused on “de-escalation” of surgical therapy. Dr. Anna Weis provided an update of clinical trials evaluating active surveillance for low-risk ductal carcinoma in-situ (DCIS): COMET, LORD and LORIS. Approximately 60,000 cases of DCIS are diagnosed annually. Patients undergoing active surveillance do not have surgery, some are treated with endocrine therapy, and all undergo regular monitoring. This is a accepted option in select cases of prostate cancer, and Dr. Weiss noted that there is no difference in overall survival in patients with low-grade DCIS who do not undergo treatment. The LORD and LORIS trials are open in the UK and the COMET study is open in the US. (Additional perspective) […]
[…] Shelley Hwang TIME Magazine 100 Most Influential People – Laura Esserman and Shelley Hwang Dr. Attai Active Surveillance for DCIS Dr. Attai DCIS Continued… Dr. Attai slideshare Are We Overtreating DCIS? DCIS411 Health News […]
Comments are closed.